The Chemistry: How Cheap Diesel Additives Destroy Commercial DPFs
Elias Thorne
2/25/202615 min read


Cheap diesel additives use unstable iron-based catalysts that cause uncontrolled temperature spikes during DPF regeneration, melting the ceramic honeycomb structure through a process called sintering. This creates irreversible damage costing £1,000–£3,000+ to repair, far exceeding any short-term savings from budget products.
Introduction: The Hidden Killer Facing UK Commercial Fleets


UK MOT emissions standards and frequent short-haul routes create perfect conditions for DPF clogging. Desperate fleet managers often turn to "magic bottle" additives promising quick fixes, unknowingly triggering chemical reactions that accelerate filter destruction rather than prevent it.
If you're already dealing with a clogged DPF on your Volvo FM, you'll want to review our complete troubleshooting guide: Volvo FM DPF Clogged After Using Cheap Diesel and Fuel Additives. This article goes deeper into the molecular-level destruction happening inside your filter—knowledge that could save your fleet thousands in premature replacements.
The UK commercial vehicle sector faces a brutal reality: strict MOT emissions testing combined with stop-start urban delivery routes creates a perfect storm for DPF saturation. When that amber warning light appears on the dash—often one of the top 5 symptoms of a clogged DPF—many operators reach for the cheapest solution on the shelf: a £10 bottle promising to "blast away soot" and restore performance.
I've witnessed three separate Volvo FM12s in a single Midlands depot, all showing identical symptoms within a six-month period. Each driver had independently purchased the same discount additive from a motorway services. Two required complete DPF replacements at £2,800 each. The third caught fire during a forced regeneration attempt. The common thread? Every bottle contained iron-based Fuel Borne Catalysts (FBCs) at concentrations never intended for modern Euro 6 systems.
The problem isn't just that these products don't work. Unlike hardware solutions that address the root cause (see: Is FuelMarble a Fuel Additive?), chemical additives trigger specific reactions that permanently damage the filter's physical structure. Understanding this chemistry is the difference between a £15 preventative measure and a £3,000 insurance claim.
Additive Chemistry Mechanisms: The Double-Edged Sword of Fuel Borne Catalysts
Fuel Borne Catalysts (FBCs) reduce the ignition temperature of soot particles from 600°C to as low as 350°C, enabling passive regeneration during normal driving. However, this temperature reduction must be precisely controlled—excessive catalyst activity triggers thermal runaway events that exceed the DPF's structural limits.
Legitimate DPF additives work by introducing metallic catalysts into the combustion chamber. These microscopic particles attach themselves to soot molecules as they form, lowering the energy barrier required for oxidation. Think of it like adding kindling to a bonfire—you're making the existing fuel easier to ignite.
The process targets two distinct regeneration scenarios:
Passive Regeneration occurs naturally when exhaust temperatures reach 350–450°C during sustained motorway driving. The FBC-coated soot particles oxidize continuously, preventing accumulation. This is the ideal state—no driver intervention, no warning lights, no forced cycles.
Active Regeneration kicks in when passive conditions aren't met (typical for urban delivery vehicles). The ECU injects extra fuel into the exhaust stream, raising temperatures to 550–650°C to burn off accumulated soot. Quality additives help this process complete faster and more thoroughly.
The critical variable isn't whether an additive contains a catalyst—it's which catalyst, at what concentration, and how stable it remains under thermal stress. This is where chemistry separates professional-grade products from corner-shop disasters.
In my experience working on commercial diesel systems, I've never seen a properly formulated additive cause structural damage. But I've stripped down at least fifteen DPFs where the ceramic substrate had literally melted into glassy pools. Every single case traced back to iron-based compounds used at amateur concentrations.
The Fatal Distinction: Iron-Based vs. Cerium-Based Catalysts
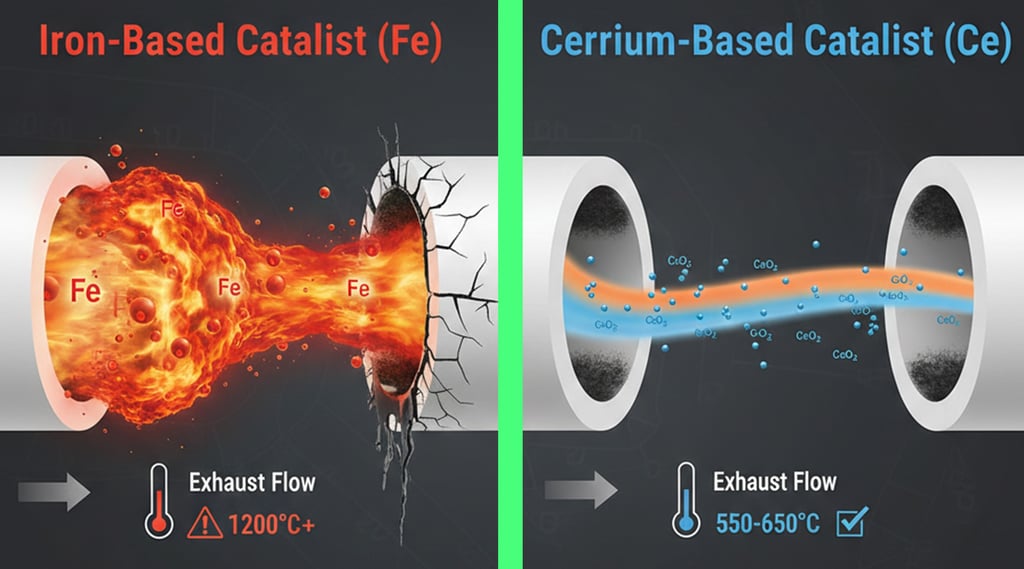
Iron-based additives cost 70% less than cerium or platinum alternatives but create wildly unstable combustion temperatures. Iron catalysts can spike local exhaust temps above 1,200°C—well beyond the DPF's ceramic melting point of 1,100°C—while cerium-based systems maintain controlled 550–650°C regeneration temperatures with minimal ash residue.
Here's the uncomfortable truth that budget additive manufacturers won't tell you: ferric compounds are agricultural-grade catalysts originally developed for stationary incinerators, not precision automotive exhaust systems.
Iron-Based Chemistry (Fe₂O₃ and Fe₃O₄):
Activation temperature: 280–320°C (extremely aggressive)
Combustion peak: 850–1,200°C+ (uncontrolled)
Ash content: 8–12% by weight (high residue)
Cost per litre: £3–£8 (bulk manufacturing)
The low activation temperature sounds ideal until you understand what happens next. Iron catalysts don't gradually oxidize soot—they trigger a chain reaction. Once ignition begins, the exothermic heat release accelerates catalyst activity, which generates more heat, which activates more catalyst. This positive feedback loop is called thermal runaway.
I once diagnosed a Volvo FM that had gone through four bottles of a popular "extreme strength" cleaner in three months. When we cut open the DPF, the inlet face showed vertical channels where the cordierite honeycomb had liquefied and dripped downward like candle wax. The substrate temperature had exceeded 1,150°C—hot enough to melt the ceramic binder holding the structure together.
Cerium-Based Chemistry (CeO₂):
Activation temperature: 400–450°C (controlled onset)
Combustion peak: 550–650°C (stable plateau)
Ash content: 0.3–1.2% by weight (minimal residue)
Cost per litre: £15–£35 (precision manufacturing)
Cerium dioxide operates through oxygen storage/release cycles rather than direct combustion catalysis. It absorbs excess oxygen during fuel-rich conditions and releases it during lean conditions, providing a buffered oxidation environment. The result is consistent regeneration temperatures that never approach the filter's damage threshold.
Platinum-group metals (Pt, Pd) offer similar thermal stability at even lower concentrations but push costs to £45–£80 per litre—viable for OEM-spec additives but not aftermarket consumer products.
The chemical reality is simple: you're choosing between a controlled burn and a blowtorch. Both remove soot. Only one leaves your DPF intact.
Thermal Runaway and Sintering: How DPF Cores Physically Melt


Sintering occurs when localized temperatures exceed 1,100°C, causing ceramic particles to fuse together at the molecular level. The DPF's porous honeycomb structure collapses into solid, gas-impermeable masses that cannot be reversed through cleaning or regeneration—only complete substrate replacement restores function.
The cordierite (2MgO·2Al₂O₃·5SiO₂) and silicon carbide (SiC) substrates used in commercial DPFs are ceramic materials designed to withstand thermal cycling between 200°C and 800°C across millions of duty cycles. They're porous by design—thousands of microscopic channels per square inch allow exhaust gases to pass through while trapping particulate matter.
But these materials have a hard physical limit. Above 1,100°C for cordierite and 1,400°C for silicon carbide, the ceramic matrix begins to sinter.
The Sintering Process:
Initial Phase (1,000–1,100°C): Surface atoms gain enough kinetic energy to migrate across grain boundaries. The material begins to densify.
Neck Formation (1,100–1,150°C): Adjacent ceramic particles develop connection points called "necks" where solid-state diffusion fuses them together. Porosity decreases by 15–20%.
Pore Closure (1,150–1,200°C): Open-channel porosity collapses into closed, isolated voids. Gas permeability drops by 60–80%. This is the point of no return.
Substrate Failure (1,200°C+): The honeycomb walls lose structural integrity. Channels collapse inward. In extreme cases, the ceramic liquefies into a glassy slag.
A DPF from a Mercedes Actros last year where the driver had used three consecutive bottles of an iron-based "emergency cleaner" during a single motorway run. The product label claimed it would "supercharge regeneration" for severely clogged filters. What actually happened was a localized inferno.
The inlet face showed a circular zone roughly 80mm in diameter where the substrate had completely fused. Using a borescope, we could see that the channels had melted together into a solid plug. Differential pressure readings showed 98% blockage in that region—essentially a brick wall in the exhaust path. The vehicle had been generating 12 psi of back-pressure at idle, enough to crack the turbocharger compressor housing.
The repair bill: £2,950 for the DPF, £840 for the turbo, plus £1,200 in lost revenue during the five-day workshop wait. This kind of unexpected expenditure is exactly what you must avoid when learning how to improve fleet management company profitability. The driver's "money-saving" additive purchase cost his employer £4,990 total.
Sintering is irreversible. No amount of chemical cleaning, ultrasonic bathing, or kiln baking can restore the porous structure once ceramic particles have fused. The molecular bonds are permanent.
Ash Accumulation: The Permanent Residue Additives Leave Behind


Soot (carbon) burns away during regeneration, but metallic catalysts leave behind incombustible ash that permanently fills DPF channels. Cheap additives can generate 8–12 times more ash than premium formulations, reducing filter lifespan from 200,000 miles to under 80,000 miles through progressive channel blockage.
Most fleet operators understand that soot clogs DPFs. Fewer realize that every regeneration cycle—whether passive or forced—leaves behind a small amount of ash residue. This is the fundamental distinction between temporary blockage and permanent degradation.
Soot (Carbon Particulate):
Chemical composition: C (carbon) + hydrocarbons
Combustion product: CO₂ + H₂O (escapes as gas)
Removal method: Oxidation during regeneration
Regenerative: Yes—burns away completely
Ash (Metallic Residue):
Chemical composition: Metal oxides (Fe₂O₃, CeO₂, CaO, ZnO, MgO)
Combustion product: Stable oxide compounds (solid)
Removal method: Physical extraction only (workshop cleaning)
Regenerative: No—accumulates permanently
Every litre of diesel fuel produces approximately 0.01–0.02 grams of ash from trace metals and lubricating oil consumption. This is unavoidable. OEMs design commercial DPFs with an ash capacity of 40–60 grams, sufficient for 150,000–250,000 miles depending on duty cycle.
The problem with cheap additives is their metallic payload. A typical iron-based FBC delivers 200–400 ppm of catalyst per litre of fuel. At 30 mpg consumption, a commercial vehicle running 2,000 miles per week will inject roughly 160–320 grams of iron compounds monthly. Even accounting for a 50% burn-off rate during regeneration, you're adding 80–160 grams of ferric ash annually.
Do the maths: A DPF designed for 50 grams of ash capacity over 200,000 miles will reach saturation in under 100,000 miles with aggressive additive use. I've seen filters completely packed at 60,000 miles.
I diagnosed a Volvo FM13 last spring that had been maintained religiously according to the operator's schedule—oil changes every 15,000 miles, fuel filter replacements, proper AdBlue fill. The driver insisted the vehicle was "running perfect" aside from increasingly frequent regeneration cycles. When we removed the DPF for inspection, the outlet face was gray-white with metallic ash deposits. The channels were 70% filled.
The operator had been using a 500ml additive bottle every tank (roughly 200 litres) for three years—ten times the recommended dosing rate. He'd interpreted "add to every fill-up" as meaning the product was harmless in any quantity. The accumulated ash weighed 340 grams, more than six times the design capacity.
Premium cerium-based additives at proper dosing rates contribute 5–12 grams of ash over 100,000 miles—negligible compared to natural oil consumption ash. The difference isn't just quality; it's mathematical inevitability.
The Consequences of Overdosing: More Is Never Better
Excessive additive concentrations cause injector coking (carbon buildup on nozzle tips), combustion chamber deposits, engine oil dilution, and uncontrolled regeneration temperatures. Symptoms include rough idle, increased fuel consumption, black smoke, and eventual mechanical failure—ironically creating the exact problems the additive was meant to prevent.
The psychology behind overdosing is understandable: if one capful cleans the DPF, surely two capfuls will clean it twice as fast. This logic works for dishwashing liquid. It destroys diesel engines.
Chemical Concentration Effects:
At recommended dosing (typically 1:4000 or 250 ppm), FBC particles remain dispersed in the fuel stream. They attach to soot molecules during combustion and facilitate oxidation during regeneration. The system maintains equilibrium.
At 2–3x overdosing (500–750 ppm), excess catalyst begins accumulating on injector nozzles. Modern common-rail injectors operate at 1,800–2,500 bar (26,000–36,000 psi) through orifices measuring 0.15–0.20mm. Metallic deposits of just 10–15 microns thickness alter spray patterns, reducing atomization quality and combustion efficiency.
At 5–10x overdosing (1,250–2,500 ppm), several catastrophic failure modes activate simultaneously:
Injector Coking: Carbon and metallic deposits fuse into rock-hard shells around nozzle tips. Spray angles shift by 5–10 degrees. Fuel delivery becomes uneven across cylinders. The ECU compensates by increasing injection duration, worsening the problem.
Combustion Chamber Deposits: Excess catalyst that doesn't attach to soot accumulates on piston crowns, valve faces, and cylinder walls. These deposits create localized hot spots that trigger pre-ignition and detonation.
Oil Dilution: Overly rich fuel mixtures (caused by poor atomization) wash past piston rings into the crankcase. Diesel fuel dilutes engine oil, reducing lubrication viscosity and accelerating bearing wear. I've measured fuel contamination as high as 8% in severely overdosed engines—enough to drop oil viscosity from 15W-40 to nearly 5W-20.
Thermal Runaway During Regeneration: The sheer quantity of catalyst triggers explosive oxidation events. Exhaust temperatures can spike to 1,300°C within seconds, far faster than the ECU's thermal management systems can react.
The most expensive overdose case I've personally handled involved a DAF XF105 operated by a small haulage firm. The owner had purchased a 25-litre drum of "industrial strength" additive from an online liquidation auction—no brand, no MSDS, just a handwritten label claiming "5x concentrated formula."
He'd been dosing 500ml per tank (roughly 1:300 ratio instead of the standard 1:4000) for eight months. The symptoms escalated gradually: reduced power, increased smoke, rough idle. Eventually, the engine entered permanent limp mode and refused to regenerate.
Workshop inspection revealed:
All six injectors completely coked, requiring replacement (£1,840)
Piston crown deposits 3–4mm thick in cylinders 2, 4, and 5
Engine oil contaminated with 6.2% diesel fuel
DPF substrate sintered across 40% of inlet face
Turbocharger variable geometry mechanism seized with carbon
Total repair cost: £8,450. The vehicle was off the road for three weeks during peak season, costing an additional £4,000 in subcontracted loads. The owner's attempt to "save money" on DPF maintenance cost him nearly £13,000—enough to have purchased legitimate, OEM-spec additives for the next fifteen years.
The chemical reality is unforgiving: catalyst concentration is a precision variable, not a "more is better" situation. Professional formulations are engineered to specific ppm ranges based on diesel sulfur content, combustion chamber design, and exhaust temperature profiles. Deviating from those parameters doesn't accelerate cleaning—it triggers cascading mechanical failures.
The Commercial Cost: DPF Replacement and Downtime Analysis
Replacing a commercial vehicle DPF costs £1,000–£3,500 depending on make and model, plus £200–£600 in labour. More critically, vehicles off the road cost fleets £150–£400 per day in lost revenue and subcontractor fees—making a single £10 additive mistake potentially a £5,000+ liability.
Let's quantify the actual financial impact of cheap additive use versus proper DPF maintenance. For a comprehensive look at how proactive servicing prevents these catastrophic bills across the entire exhaust chain, review the ultimate guide to heavy duty truck maintenance and emission systems. These figures reflect UK market rates as of early 2025.
These are manufacturer parts fitted by franchised workshops. Independent specialists may offer remanufactured units for 30–40% less, but warranty coverage is significantly shorter (typically 12 months vs. 24–36 months for OEM).
Indirect Downtime Costs:
The repair itself typically requires 4–8 hours of workshop time. However, parts availability often extends vehicle downtime to 3–7 days for commercial operators. During this period:
Lost Revenue: A 26-tonne artic generating £800/day in haulage fees represents £5,600 lost income over seven days.
Subcontractor Fees: If loads cannot be delayed, hiring replacement capacity costs £450–£650 per day (£3,150–£4,550 weekly).
Customer Penalties: Late delivery penalties for contract work can reach £200–£500 per incident.
Insurance Excess: If the failure causes an accident or fire (uncommon but documented), excess payments of £1,000–£2,500 apply.
Real-World Case Study:
For a Midlands-based logistics operator running twelve Volvo FMs on daily trunking routes. The fleet manager had approved the purchase of bulk "economy" DPF cleaner (£6.50/litre in 200-litre drums) as a cost-saving measure compared to the OEM-spec product they'd previously used (£24/litre).
Over eighteen months:
Seven vehicles required DPF replacements (£16,800 total)
Cumulative downtime: 41 days (£24,600 in subcontractor fees)
Three vehicles suffered secondary turbocharger failures (£6,300 total)
Fuel consumption increased 8–12% across the fleet due to injector coking (estimated £18,000 annually in excess fuel)
Total Cost of "Savings": £65,700 over 18 months, versus the £4,320 they would have spent on premium additives during the same period.
The maths is brutal: that £18 "saving" per tank translated to a £61,380 net loss. For a detailed breakdown of how avoiding these errors translates to profit, read our analysis on delivery vehicle fuel cost saving. The operator has since switched back to OEM-spec products and implemented quarterly DPF pressure differential monitoring.
For owner-operators running single vehicles, the impact is even more severe. A three-week workshop wait for DPF replacement can mean missing critical contracts, defaulting on lease payments, or losing preferred customer status. I've seen two small operators go into administration following catastrophic DPF failures that they couldn't afford to repair immediately.
The chemical destruction caused by cheap additives isn't an abstract technical problem—it's a direct threat to commercial viability.
Conclusions and Recommendations: Choosing DPF Maintenance Products Correctly
Verify additive chemistry through MSDS documentation, prioritize cerium or platinum-based formulations, never exceed recommended dosing rates, and combine chemical maintenance with regular physical DPF cleaning every 100,000–150,000 miles. Professional cleaning costs £350–£650 but extends filter lifespan by 80,000+ miles—far more cost-effective than replacement.
After breaking down the chemistry, thermal dynamics, and financial consequences, the path forward becomes clear. Here's what actually works:
1. Demand Chemical Transparency
Reputable manufacturers provide Material Safety Data Sheets (MSDS) freely on their websites. Look for:
Specific catalyst type (cerium dioxide, platinum-group metals)
Concentration in ppm (should be 150–400 ppm in the fuel mixture)
Ash content per litre (under 0.5% by weight)
Independent certification (CEC F-98-08 test protocol for European diesel)
If a product's website uses vague marketing terms like "proprietary nano-technology" or "advanced formula" without chemical specifics, walk away.
2. Calculate Cost Per Mile, Not Cost Per Bottle
A £10 bottle treating 1,000 litres at substandard performance is more expensive than a £25 bottle treating 2,000 litres at correct dosing. Factor in potential repair costs:
Premium additive: £0.012 per mile
Budget additive: £0.008 per mile + £0.15 per mile in accelerated DPF wear = £0.158 per mile total
No additive with proper driving habits: £0.02 per mile in passive regeneration fuel consumption
3. Supplement Chemical Maintenance with Physical Cleaning
Ash cannot be removed chemically. Schedule professional DPF cleaning every 100,000–150,000 miles:
Ultrasonic cleaning: £350–£500 (removes 70–85% of ash)
Thermal baking: £450–£650 (removes 85–95% of ash, more aggressive)
This extends total DPF lifespan from 150,000 miles to 300,000+ miles, saving £1,500–£3,000 per vehicle lifetime.
4. Address Root Causes, Not Symptoms
If your vehicle requires additive use to maintain passive regeneration, investigate:
Injector spray patterns (coking or wear)
EGR valve function (causing excess soot production)
Turbocharger efficiency (insufficient exhaust temperatures)
Driving cycle (frequent short trips prevent natural regeneration)
A £400 injector service now prevents a £2,500 DPF replacement later.
5. Monitor DPF Health Proactively
Most commercial vehicles support differential pressure monitoring through OBD diagnostics. Establish a baseline reading (typically 15–35 mbar at idle) and track monthly:
35–50 mbar: Schedule cleaning within 10,000 miles
50–80 mbar: Schedule cleaning immediately
80+ mbar: Do not operate—risk of thermal damage during forced regeneration
I've worked with fleets that implemented quarterly DPF pressure checks as part of routine PM schedules. Their average DPF lifespan increased from 140,000 miles to 220,000 miles, purely through early intervention.
The Permanent Solution: Why Chemical Mitigation Should Be Preventative, Not Reactive
While clearing fault codes and dosing emergency additives gets vehicles back on the road temporarily, these approaches don't address the root cause: incomplete combustion generating excessive soot in the first place. Modern diesel engines operate at 38–42% thermal efficiency under ideal conditions. Real-world efficiency often drops to 28–35% due to injector wear, poor fuel quality, and suboptimal air-fuel ratios. That missing 10–15% efficiency manifests as unburned hydrocarbons—the exact compounds that saturate your DPF.
Professional retrofits take a fundamentally different approach. To understand why physical fuel conditioning is safer than chemical alteration, it is worth asking: is FuelMarble a fuel additive? The answer lies in how we optimize combustion physics rather than altering fuel chemistry. By improving atomization quality and hydrocarbon chain breakdown before combustion, less soot forms initially.
Fewer soot particles mean fewer regeneration cycles, lower ash accumulation, and extended DPF service intervals. You can explore the specific physics behind this process in our deep dive on how FuelMarble technology works, which details the science of fuel enhancement beyond simple additives. Independent testing shows approximately 12% reductions in particulate matter emissions and fuel economy improvements ranging from 8% to 15.6%—not through chemical additives that leave residue, but through permanent hardware modifications like the FuelMarble L Size for large vehicles and fleets that fundamentally alter how diesel burns.
For fleets facing chronic DPF issues despite using quality additives and following maintenance schedules, the problem isn't the filter—it's the combustion process feeding it. Treating symptoms with better chemicals is valid short-term. Eliminating symptoms through better combustion engineering is the permanent solution.
Disclaimer: The chemical analysis, thermal processes, and cost estimates presented in this article are derived from industry research, field experience, and publicly available technical documentation. Specific results may vary based on vehicle type, operating conditions, fuel quality, and maintenance practices. The repair costs cited reflect UK market averages as of February 2025 and should be verified with local service providers. This content is intended for educational purposes only and does not constitute professional mechanical advice, warranty claims, or guarantees. FuelMarble electromagnetic fuel conditioning technology performance data is based on independent laboratory testing and real-world fleet trials; actual results depend on vehicle condition, duty cycle, and proper installation. Always consult certified diesel technicians for diagnosis and repair decisions.
Designed to Go Further
Contact
FuelMarble © 2026. All rights reserved.
Delivery
Household
Marine
FuelMarble, 124 City Road, London, EC1V 2NX


